Fourth Generation Cancer Immunotherapy NKT Cell Targeted Therapy
Description of RIKNKT® Using AlphaGalactosylceramide-Stimulated Autologous Dendritic Cells
This treatment has been deliberated and approved by the Japan Society for Regulatory Science (JSRS) Committee on Regenerative Medicine, etc., based on the examination work specified in the Act on Securing Safety of Regenerative Medicine, etc. (Act No. 85 of 2013) and Ministry of Health, Labour and Welfare Ordinance No. 140. A provision plan has been submitted to the Minister of Health, Labour and Welfare.
Name of Regenerative Medicine, etc.: Alpha Galactosylceramide-Stimulated Autologous Dendritic Cell NKT Cell Targeted Therapy RIKNKT® Medical Institution Providing Regenerative Medicine, etc.: Solaria Clinic Tokyo Facility Manager: Yoshiatsu Koga Person Responsible for Implementation and Physician Performing Regenerative Medicine, etc.: Yoshiatsu Koga
- Contents of NKT Cell Targeted Therapy
1) What is Immunocellular Therapy
Current standard cancer treatments consist of surgery, chemotherapy, and radiation. However, in cases where these standard treatments are not feasible due to side effects or physical conditions, or in cases of metastasis or recurrence, treatment becomes more challenging. Therefore, attempts have been made for over 30 years to apply immune cells that attack cancer to cancer treatment, and with the rapid advancement of immunology, clinical trials of new cancer immunotherapies are being conducted worldwide.
2) Types of Immunocellular Therapy
Cancer immunotherapy includes the use of activated αβ or γδ T lymphocytes, cancer peptide vaccines, dendritic cell vaccines, NK (Natural Killer) cells, NKT (Natural Killer T) cells, as well as immunotherapy using genetically modified immune cells. Among these, some have already been approved, some are under research or clinical trials, and some are provided as self-pay treatments in private medical institutions.
3) What is NKT Cell Targeted Therapy (RIKNKT®)
The immune system that defends the body consists of “innate immunity” and “acquired immunity.” “Innate immunity” is responsible for initial defense by sensing various antigens (invading pathogens or abnormal self-cells) and eliminating them, while “acquired immunity” distinguishes antigens specifically and effectively eliminates them by memorizing them. Immune cells responsible for these functions are primarily NK cells for the former and T cells for the latter. NKT cells have characteristics of both NK cells and T cells, hence the name. In NKT Cell Targeted Therapy (RIKNKT®), monocytes in the patient’s blood are collected, differentiated into dendritic cells, and stimulated by “GMP-compliant Alpha-Galactosylceramide” to activate dendritic cells. When these dendritic cells are reintroduced into the body, they activate NKT cells. NKT cells themselves have cytotoxicity against cancer cells, and additionally, activated NKT cells activate NK cells and T cells, exerting potent immune cytotoxic effects against cancer cells. This method has been researched at the RIKEN Institute and Chiba University, and clinical trials are underway. Moreover, the “GMP-compliant Alpha-Galactosylceramide” used in our clinic is manufactured to high safety standards (developed by RIKEN Institute for Immunology Regenerative Medicine, and under an exclusive supply contract with manufacturing commissioned companies, with no other suppliers).
※GMP standards refer to regulations stipulated under the law titled “Ordinance for Standards for Manufacturing and Quality Control of Pharmaceuticals and Quasi-drugs,” which outlines the requirements that manufacturers of pharmaceuticals must adhere to. “GMP” is an abbreviation for “Good Manufacturing Practice” and refers to the standards set forth in the “Ordinance for Standards for Manufacturing and Quality Control of Pharmaceuticals and Quasi-drugs.”
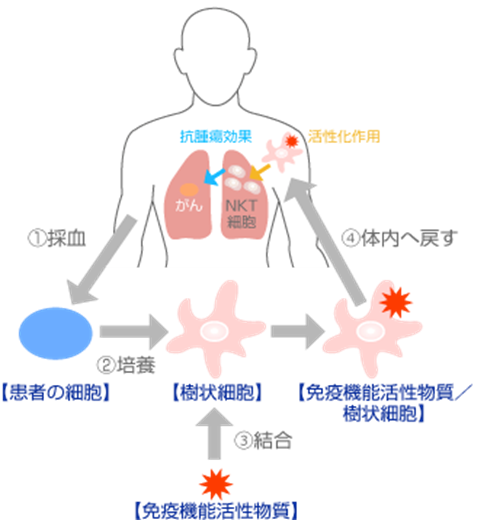
Reference: RIKEN Center for Integrative Medical Sciences
4) Indications
Individuals with malignant tumors or those requiring improvement in immune function as determined by a physician.
5) Eligibility Criteria
① Gender: Any
② Age: 16 years and older
③ ECOG Performance Status: Within the range of 0 to 3.
④ Consent:
i. Agreeing to the contents outlined in the “Explanation and Consent Form for ‘NKT Cell-Targeted Therapy Using Alpha-Galactosylceramide-Stimulated Autologous Dendritic Cells (RIKNKT®)'”. ii. Agreeing to the contents outlined in the “Request for Research Use and Consent Form”.
⑤ Exclusion Criteria and Excluded Conditions:
i. Patients with T-cell or NK-cell type malignant lymphoma or leukemia who do not consent to the same treatment. ii. Individuals with severe autoimmune diseases (such as psoriasis) deemed ineligible for application by a physician. iii. Active bronchial asthma. iv. Bacterial infections (diagnosed based on symptoms such as fever, leukocytosis). v. History of bone marrow or organ transplantation. vi. Pregnant or potentially pregnant women, as well as breastfeeding women. vii. Carriers of viral infections such as HIV, HTLV-1, or individuals suspected of syphilis infection. viii. Complications of severe cardiac, pulmonary, hepatic diseases, infectious diseases, as well as diseases and conditions judged unsuitable by a physician considering comprehensive factors such as liver function, renal function, and malnutrition. Specific numerical criteria deemed unsuitable include:
- White blood cell count: 10,000/uL or higher
- Hemoglobin: Less than 9.0g/dL
- Platelet count: Less than 100,000/mL
- Blood pressure: Systolic blood pressure of 200mmHg or higher or 80mmHg or lower
- SpO2 (room air): Less than 94%.
6) Procedure from Blood Collection to Cell Administration
① We conduct interviews, examinations, and tests for infectious diseases. The results of infectious disease tests will be communicated only to the patient themselves and individuals specified by the patient. If tests for HIV, adult T-cell leukemia virus, or syphilis are positive, the patient may not undergo NKT cell-targeted therapy due to the potential worsening of the infection or priority given to treating the infection.
Testing Content
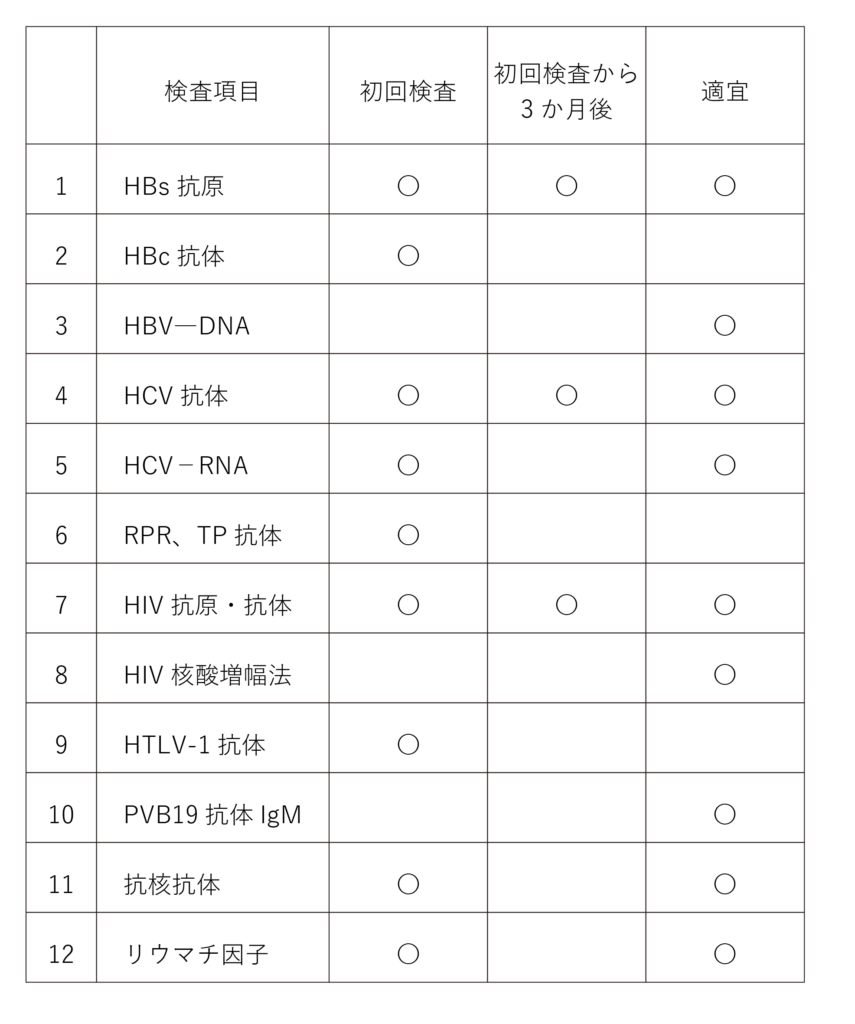
The initial examination is a prerequisite for receiving cell therapy. The examination conducted three months after the initial examination is to address the “window period” where viruses may not be detected immediately after infection. Please undergo the examination three months later whenever possible. Additionally, examinations conducted as needed are performed clinically, such as after transfusions.
② If it is determined that treatment is feasible based on the results of ①, a method called apheresis, which involves collecting a large amount of monocytes from the blood, is performed. Apheresis is necessary because monocytes, which are the precursor cells of dendritic cells, comprise less than 10% of white blood cells. This procedure utilizes specialized medical equipment for apheresis, and approximately 4000 mL of blood is circulated externally. Monocytes are then collected, typically taking 2 to 3 hours. Specifically, an injection needle is inserted into a vein in the arm or groin area and connected to the machine to selectively collect monocytes, while the rest of the blood is returned to the bloodstream. In rare cases, if the physician determines that component collection cannot be performed from the vein due to the condition of the patient’s blood vessels, component collection may be performed from the artery.
③ Monocytes collected from the blood are differentiated into dendritic cells and then cultured for a total of 8 days, primarily stimulated with “GMP-compliant alpha-galactosylceramide.” Cell culture is outsourced to the Cell Culture Processing Facility at BioAxel Co., Ltd. Kyoto University Katsura Venture Plaza.
④ After culturing is complete, dendritic cells stimulated with “GMP-compliant alpha-galactosylceramide” are frozen and stored at the Cell Culture Processing Facility at BioAxel Co., Ltd. Kyoto University Katsura Venture Plaza.
⑤ To confirm the safety of the frozen dendritic cells, infection tests (such as sterile tests) are conducted over a period of one week.
⑥ After confirming safety in ⑤, the frozen dendritic cells stimulated with “GMP-compliant alpha-galactosylceramide” are adjusted, and typically administered four times every two weeks. However, this may vary if deemed appropriate by the physician and with the patient’s consent. Administration is performed via intravenous drip infusion, typically mixed with approximately 50 mL of saline solution.
7) Ensuring the Safety of Administered Cells
Culturing of dendritic cells is conducted under strict technical supervision at the Cell Culture Processing Facility at BioAxel Co., Ltd. Kyoto University Katsura Venture Plaza. However, it is crucial to confirm multiple times before administration that cultured cells are not contaminated with bacteria or other microorganisms. To ensure the safety of cell administration, sterile tests, mycoplasma negative tests, endotoxin tests, etc., are conducted on cultured cells (dendritic cells stimulated with “GMP-compliant alpha-galactosylceramide”) before administration to confirm their safety.
8) In Case Cultured Dendritic Cell Counts Do Not Reach Specified Levels
Culturing of dendritic cells is carried out under strict technical supervision in a cleanroom. However, the characteristics and quantity of dendritic cells obtained may vary depending on the condition of the collected blood, affecting cell separation, recovery, and adjustment. If an insufficient number of dendritic cells can be obtained or if they fail the tests, treatment cannot proceed as scheduled.
9) Provision and Storage of Blood and Cultured Cell Samples Blood and cultured cells used for treatment are required to be stored for a certain period as reference samples in case of infections, etc. A small portion of the patient’s blood and cultured cells will be provided. The amount of stored samples is only a small fraction (approximately 1 mL) of the quantity required for treatment and will not affect the treatment process.
The provided samples will be stored and managed for one year at the Cell Culture Processing Facility at BioAxel Co., Ltd. Kyoto University Katsura Venture Plaza. If diseases or other occurrences arise during or after treatment, the stored samples will be used for investigation purposes. The stored samples for investigation purposes will be kept frozen. If not used, they will be appropriately disposed of one year after storage commencement. The provided samples will only be used for the disclosed purposes and will not be used for the treatment of others.
- Expected Effects and Side Effects
1) Regarding the effects
Clinical trials of NKT cell-targeted therapy have already been conducted at universities and other research institutions, with results suggesting significant efficacy in advanced lung cancer and head and neck cancer. However, please note that the effects of treatment may vary depending on individual patient conditions such as disease status, pathology, and blood condition. Therefore, please be aware of potential differences among patients. In our clinic, we aim to collect and verify necessary examination data to assess effectiveness by collaborating with the hospitals where patients are currently receiving treatment or consultation. We will continue to provide reliable treatment tailored to each patient to ensure optimal results. While theoretically, NKT cell-targeted therapy is a treatment that could be applicable to all cancer patients, it has not been validated as a treatment method for all cancer patients. As a reference, we present the results of physician-led trials of NKT cell-targeted therapy for lung cancer and head and neck cancer.
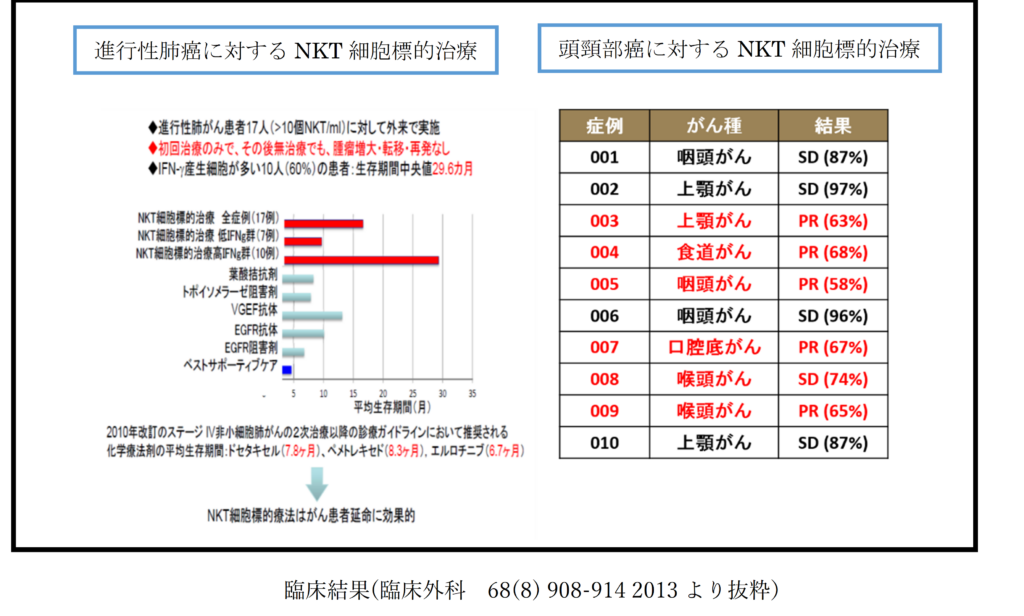
2) Regarding treatment side effects
After administering dendritic cells stimulated with “GMP-compliant alpha-galactosylceramide,” mild fever and fatigue may occur, but in most cases, these are minor, with fever typically being below 38°C and resolving within two days. Additionally, very rarely, symptoms suggestive of allergic reactions may occur. We will conduct treatment with careful observation, and if such side effects occur, we will promptly and appropriately address them.
3) Regarding side effects associated with apheresis
Blood is circulated externally from blood vessels such as the arm or groin, with punctures performed by a proficient physician after thorough disinfection. Occasionally, subcutaneous bleeding may occur. Additionally, due to tension from blood collection, dizziness, nausea, and even hypotension may occur, but adjusting the collection rate while observing symptoms helps mitigate their occurrence. Anticoagulants are used during apheresis to prevent blood coagulation, which may lower blood calcium levels due to the citrate contained in them. Consequently, initial symptoms such as tingling in the lips and fingers may occur, progressing to nausea, vomiting, and muscle twitching in the fingers. If initial symptoms are observed, the collection or return rate may be slowed at that stage, or corrective medication to normalize calcium levels may be administered to improve symptoms. Occasionally, thrombocytopenia may be observed, but since it quickly recovers, platelet transfusion is not necessary.
- Availability, Content, Comparison with Effects and Side Effects of Other Treatment Options
1) Other Treatment Options
There are three standard cancer treatments: surgical procedures, chemotherapy, and radiation therapy. Additionally, there are new antibody therapies available recently. These treatment methods have already been established as cancer therapies. If undergoing or planning to undergo these treatments, we will determine the optimal timing and intervals for administering dendritic cells stimulated with “GMP-compliant alpha-galactosylceramide” according to the treatment schedule as much as possible. - Comparison with the Effects of Other Treatment Methods
Standard treatments such as surgical procedures, radiation therapy, and chemotherapy have been extensively studied and applied clinically both domestically and internationally for many years, providing scientific and medical evidence of their effectiveness in tumor reduction and extending lifespan.
NKT cell-targeted therapy (RIKNKT®), developed by RIKEN Immunotherapy, is currently being legally provided in compliance with the Act on the Safety of Regenerative Medicine (Act No. 85 of 2013) established by the Ministry of Health, Labour and Welfare.
- Comparison with side effects of other treatments
The side effects of standard treatments, including their nature, severity, and frequency, are well documented. They range from mild symptoms such as loss of appetite, diarrhea, hair loss, and skin disorders to more severe conditions like peripheral neuropathy and bone marrow suppression. In contrast, such side effects are rarely observed with our therapy. - Post-treatment follow-up survey
Under the Regenerative Medicine Law (Law No. 85 of Heisei 25), a certain period of follow-up survey is mandated for monitoring the progression of the patient’s condition and any occurrence of diseases after the treatment. In order to track the presence of side effects and the effectiveness of the treatment, we will send you a follow-up survey form from our clinic to the contact information provided, for a period of 5 years after the completion of NKT cell-targeted therapy (RIKNKT®). We kindly ask for your cooperation. - Refusal is voluntary
Even if you have consented to this treatment, you have the right to withdraw your consent and discontinue the treatment at any time, regardless of whether it is before or after the start of treatment or before or after the blood collection for culturing. Revoking your consent will not result in any disadvantage in subsequent treatments. However, if you withdraw your consent after the start of cell culturing, the cell culturing fee as stipulated will be charged. - Protection of Personal Information
We protect personal information based on our clinic’s personal information handling regulations. Privacy-related information such as names that can identify patients will not be provided to or disclosed to others without the patient’s consent.
Personal information such as names will be strictly managed for cell culturing and preservation by BioAxcel Co., Ltd., Kyoto University Katsura Venture Plaza Cell Culture Processing Facility, and other related facilities, as well as by RIKEN Immunology Frontier Research Center, the provider of “GMP-compliant alpha-galactosylceramide,” which manages the treatment process. Information sharing ensures strict management of cell culturing, storage handling, and cell management.
- Costs Associated with the Procedure
Initial Consultation Fee: ¥30,000 (excluding tax)
Infectious Disease Testing Fee (only for initial consultation): ¥30,000 (excluding tax)
Treatment Fee: ¥3,145,000 (excluding tax)
*In the event of withdrawal of consent or for any other reason, charges for cell culturing incurred
up to the point of initiation will be billed.
10.Compensation for Health Damage
In the event of health damage occurring to the patient due to blood collection or treatment, appropriate measures will be taken by the clinic if it can be managed there. If management is difficult, the patient will be referred to a hospital where hospitalization is possible for treatment. In cases where compensation for health damage deemed to be due to negligence is necessary, compensation will be provided through medical professional liability insurance for the physician or medical facility.
11.Ownership of Patent Rights, Copyrights, Property Rights, and Economic Interests
As a result of treatment, patent rights or economic interests related to the treatment may arise. However, since these are insights obtained from the entire provided sample, individual patients do not have the right to patent rights, copyrights, property rights, or economic interests.
12.Freedom to Ask Questions
For inquiries or complaints regarding the provision, preservation, or research of this treatment, please contact Solari Clinic physicians (open from 11:00 AM to 6:00 PM; closed on Sundays).
13.About the Certified Regenerative Medicine Committee
The NKT Cell Targeted Therapy (RIKNKT®), utilizing Alpha-Galactosylceramide-stimulated autologous dendritic cells, provided at Solari Clinic Tokyo, is undergoing evaluation by the Certified Regenerative Medicine Committee of the Japan Pharmaceutical Law and Jurisprudence Society.
Japanese Pharmaceutical Law and Jurisprudence Society Regenerative Medicine Committee (Certification Number: NB3140007) Contact Information: OGURA Building 6F, 8-18-11 Ginza, Chuo-ku, Tokyo Phone: 03-6264-3883 | Email: info@japal.org Contact Person: Dr. Yoshitsugu Koga Phone: 03-6665-6377 | Fax: 03-6665-6378


